BACKGROUND – INDUSTRY GOLD STANDARD
Double programming is widely used in the industry to improve the quality of output produced in the reporting of the results of clinical trials. In our recent survey, 100% of the 22 companies responding, both biopharmaceutical companies and CROs, responded that they use double programming, at least for some of the outputs. This approach is useful for individual tables, but it does not compare the output across tables.
THE CHALLENGE
Within an individual table, the two programs may produce the same answer, but it may not be correct. Comparing the results across tables may identify mistakes in one of the tables. For example, the number of subjects in the safety population in the disposition table may be different from the number of subjects shown in a specific analysis of the safety population, such as adverse events. This is something we have seen in our analyses of many tables. Another example where we have seen errors is when we compare the number of adverse events causing withdrawal with the number of subjects withdrawn because of an adverse event. To overcome this limitation of double programming, statisticians perform a manual check for consistency across tables. While this will improve quality, there are shortcomings.
SOLUTION
Verify, a SAAS Solution for table data validation, is a machine learning (ML) based tool. In addition to performing within table checks, can perform cross table checks quickly and consistently for all deliverables. This would be achieved by running a set of standard cross table checks defined by statisticians.
Example from PhUSE 2021 session
The impact of inconsistency:
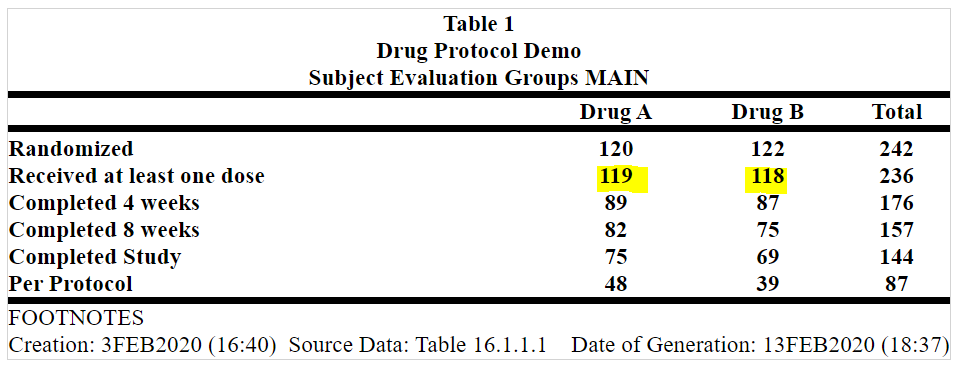
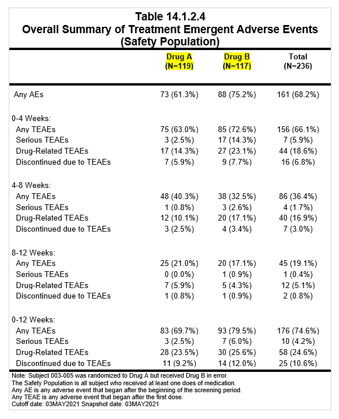
The key to success in implementing automation is the combination of the human factor and artificial intelligence (AI) or ML. By adopting an automated platform, pharma companies and CROs can:
- Greatly reduce the time and effort to perform across table checks
- Check the output comprehensively and consistently
- Improve job satisfaction
- Improve the productivity of statisticians
